Coating action and basic method introduction
There are two main reasons why electrical connectors should be coated. The first reason is to contact with the base material of the shrapnel plays an anti-corrosion protection role; The second reason is to optimize the properties of the contact interface to improve the electrical and mechanical properties of the connector.


Consider corrosion prevention first
Most connector contact shrapnel is copper alloy material, in the typical connector use environment will be subjected to corrosion, such as oxidation or vulcanization corrosion. The use of contact coating effectively blocks the direct contact between the contact shrapnel and the environment, so as to prevent copper corrosion.
Of course, the coating material in the service environment should not produce corrosion, at least not harmful corrosion. Since corrosion prevention is an important function, the selection of suitable contact coatings is a major consideration in the process of contact interface optimization.
Among the mechanical properties of greatest concern are durability, wear of coatings, and effects on fit.
These considerations are based on the effect of the separation movement of the cold welded connection between the contacts of the contact surfaces in the relative movement of the connector.
The most important mechanical properties include the hardness, durability and coefficient of friction of the coating material. These properties depend on the inherent properties of the coating material and the process by which it is applied.


Optimizing the electrical performance of the connector can start from considering the management of the plating layer, which already exists on the surface of the contact plating layer (oxide layer, etc.) and the film layer that may be formed. The primary electrical performance requirement of a connector is to establish and maintain stable contact resistance.
In order to provide a stable connection, a metal contact interface must be formed. To establish a metal contact interface, it is necessary to prevent the formation of the contact surface film or to remove the surface film. The way these two manage the layers defines the difference between precious and non-precious metals in the coating material.


Precious metal coatings (gold, palladium, and alloys of these metals) inherently do not form various surface coatings. For this type of surface treatment, the creation of the metal interface is relatively simple, as it is only necessary to remove surface contamination during the mating process. Usually this will be easy.
To maintain a stable contact impedance, connector design requirements focus on keeping the precious metal contact surface free from external factors such as contamination, diffusion of substrate metal, and contact wear.
Ordinary metal coatings, typically tin, tin-lead alloys, etc., will be covered with an oxide film. The usefulness of tin as a contact coating is based on the fact that its oxide film is easily punctured and metal contacts are easily established.
For tin-plated contacts, the immediate requirements of the connector design are to pierce the oxide film during mating and to ensure that the contact interface does not re-oxidize during the life of the connector.
The re-oxidation process in the form of fretting corrosion is the main failure mechanism of tin coatings. Silver coatings are best considered non-precious metal coatings because silver coatings tend to form oxides and sulfides. Nickel plating is also a non-precious metal fault, again due to the tendency to form oxides.


Coating method
Contact plating can be achieved in many ways, the three main plating methods are:
❉ Electroplating
❉ Spray plating
❉ Hot dip galvanizing
Electroplating process
In connector manufacturing, electroplating is the most commonly used electroplating method.
Figure 1 shows a typical electroplating tank structure. The plating layer will be deposited on the cathode (the connector is the contact shrapnel), and the metal ions will come from the plating solution, through the composition of the plating solution, or through the decomposition of the anode.
When the deposition occurs, the anode will continue to decompose and replenish the plating solution. . Inside this simple electroplating bath, the deposition process is controlled by the bath chemistry and the current distribution on the cathode surface.
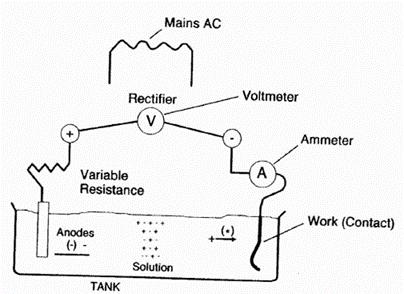

Figure 1 Schematic diagram of the electroplating tank
The phenomenological description of the electrodeposition process is simple in principle. In the case of gold plating, the plating material is deposited at various points on the metal substrate and grows laterally across the connector surface as the plating thickness increases.
When the thickness reaches a certain level, the coating will completely cover the substrate. The extent and degree of complete coverage depends on the characteristics and cleanliness of the substrate surface, as well as the plating process. The most common plating defect in electroplating is the presence of plated holes.


Most connector electroplating is done in a continuous roll-to-roll process to take advantage of the cost-effectiveness of this process. In the 1980s and early 1990s, due to the high price of gold, about 800 US dollars, a lot of resources were invested in reducing the thickness of the gold coating, and finally achieved great progress.
The thickness of the gold plating is made possible; on the one hand by controlling the gold plating location and the number of contact areas that need to be gold plated.
There are three ways to contact the plating: full plating, selective plating, and double plating. Full plating is to cover all contact areas with plating. Tin plating is generally a full-coverage plating. For precious metal coatings, considering the cost factor, the selection method of the coating generally adopts the selective plating or double-layer plating method.
In both methods, a precious metal coating is applied to the separable contacts. Selective gold plating is also used on the permanent connection end, but the thickness of the plating on both ends may be different. Typically, tin or tin-lead alloys are used for double-layer electroplating on permanent connections.


Figure 2 Classification of plating methods:
(a) full plating
(b) selective plating
(c) double-layer plating
It should be noted that for the same metal material, the material properties after electroplating are completely different from those of forged materials, especially for precious metal coating materials.
Generally, after the material is electroplated, the hardness increases, the ductility decreases, and the density decreases compared to the forging method. The magnitude of the change depends on the material and the way it is plated.


Sputtering process
Sputtering is to attach the plated metal to the base metal surface under high pressure. There are three common spray plating methods: full plating, spot plating, and embedded plating. Full plating is where the plated metal completely covers the substrate.
Spot plating only selectively covers the substrate where the coating is needed. Insert plating is a special spray plating method, which needs to cover the plating material on the recessed position of the substrate.
Hot dip galvanizing
In connector applications, hot-dip plating is limited to tin and tin alloy coatings. Hot dipping is the process of drawing strip metal through a pool of molten tin to coat it with tin. The control of coating thickness is controlled by the process, including air knife and brush. Therefore, typical tin plating thicknesses and specific thickness requirements are process technology dependent.


From the point of view of the contact interface, the main difference between hot dipping and electroplating is that the formation of intermetallic compounds is formed during the immersion process. Even at room temperature, the formation of metal compounds between copper and tin is also very easy. If you are not careful, or if the process level is relatively poor, a large amount of metal compounds will be generated during the hot dip plating process.
Excessive intermetallic compounds will have an unacceptable effect on the contact interface and will also negatively affect the solderability between the contact interfaces. Since metal compounds are produced during hot dip plating, the process must be tightly controlled to ensure that the surface coating is tin and not metal compounds.


The above is the introduction of the coating function and the basic method. For more information on connectors, please check our official website: http://www.lhecn.com/








What’s Taking place i am new to this, I stumbled upon this I’ve found
It positively helpful and it has helped me out loads.
I am hoping to contribute & help different customers
like its aided me. Great job.